 Along with these techniques, several process analytical technologies (PATs) available for product and process characterization have been summarized in literature. Even though SEC employs mild isocratic and elution conditions that confer minimal impact on the conformational stability, protein aggregates with weak intermolecular affinity can dissociate into monomers in the mobile phase. Although some ultra high-performance liquid chromatographic (UHPLC) methods can have shorter run times, method development and validation for each product and different formulations are time consuming. However, the chromatographic methods are destructive techniques which typically require long equilibration and analysis times, while there is a requirement for additional reagents and consumables, including buffers and columns. Currently, UV–Vis spectroscopy and chromatographic methods including reversed-phase high-performance liquid chromatography (RP-HPLC) and size exclusion chromatography (SEC) are the quality control (QC) release tests for concentration, purity, aggregation and degradation. However, the drawbacks associated with these methods include the requirement for additional reagents, sample preparation, compatibility with sample type, interference from multiple absorbing species and analysis time. Historically, several methods such as Bradford protein assay, bicinchoninic acid (BCA), Lowry’s assay and other dye-based methods have been used to assess protein concentration. To ensure the safety, quality and efficacy of these products as per the International Council for Harmonisation (ICH) guidelines and current good manufacturing practices (cGMP), various analytical and characterization techniques are employed prior to batch release. 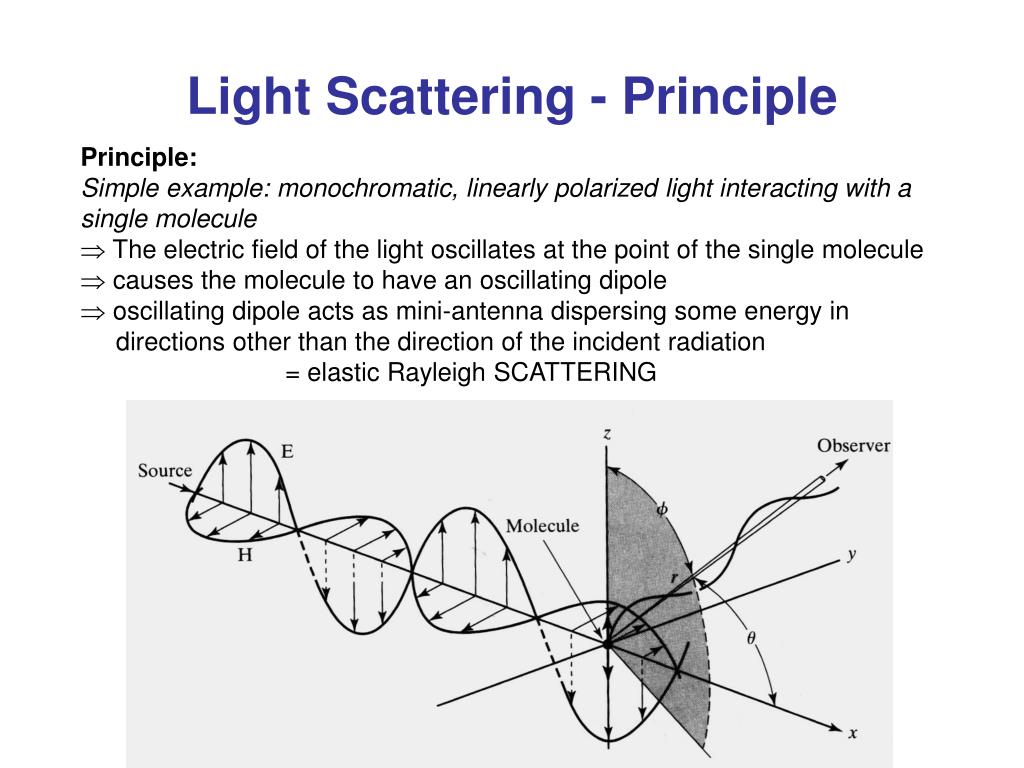
Protein aggregation is one of the critical quality attributes (CQAs) that can lead to increased immunogenicity and decreased product efficacy though in some cases dimerization and oligomerization can be integral to protein activity. It is well-known that proteins are sensitive molecules and susceptible to physical and chemical instability resulting in aggregation, denaturation, etc. The obtained calibration curve ( R 2 > 0.95) for the particle number concentration of the 3 proteins and the observed correlation between MADLS and SEC ( R 2 = 0.9938) for the analysis of aggregation in the enzyme can be employed as a 3-in-1 approach to assessing particle size, concentration and aggregation for the screening and development of products while also reducing the number of samples and experiments required for analysis prior to other orthogonal tests. In this study, the application of multi-angle dynamic light scattering (MADLS) was evaluated for the determination of particle size, particle concentration and aggregation in 3 different protein modalities, namely bovine serum albumin (BSA) and two biopharmaceuticals including a monoclonal antibody (mAb) and an enzyme. 
However, other techniques such as dynamic light scattering (DLS) are employed in tandem to measure the particle size distribution (PSD) and polydispersity of biopharmaceutical formulations. Size exclusion chromatography (SEC) is the gold standard to measure protein aggregation and degradation. To ensure their safety, quality and efficacy, a wide range of critical quality attributes (CQAs) such as product concentration, aggregation, particle size, purity and turbidity have to be met. Biopharmaceuticals are large, complex and labile therapeutic molecules prone to instability due to various factors during manufacturing. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
